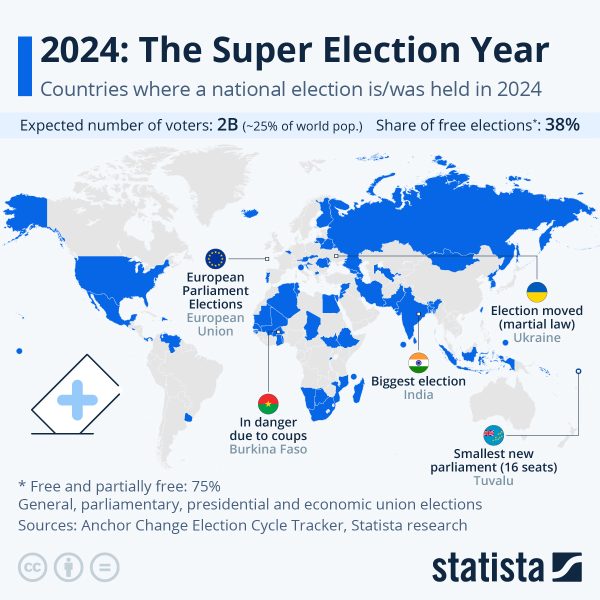
With the US reaching a peak of Covid-19 cases at about 150,000 cases a day, Pfizer, one of America’s largest pharmaceutical companies, held a press conference regarding their seemingly effective vaccine. They say that they have had a 90% success rate. In order for this vaccine to be available to the public, the FDA requires 2 months of safety data, which Pfizer will approach next week. They began phase 3 of their vaccine in July, and they have enrolled approximately 43,538 patients.
With the US reaching a peak of Covid-19 cases at about 150,000 cases a day, Pfizer, one of America’s largest pharmaceutical companies, held a press conference regarding their seemingly effective vaccine. They say that they have had a 90% success rate. In order for this vaccine to be available to the public, the FDA requires 2 months of safety data, which Pfizer will approach next week. They began phase 3 of their vaccine in July, and they have enrolled approximately 43,538 patients. Experts say that there will likely be a lower success rate in the real world because of so many different kinds of conditions and environments people live in. Clinical trials are conducted in a controlled setting, unlike in the real world. Pfizer has not yet released if there have been any side effects or if the vaccine affects the elderly differently than the younger population.
Now, we can’t get too ahead of ourselves. Just because the vaccine may be very effective, doesn’t mean that we won’t need to take any precautions anymore. We will still need to wear masks in public and maintain social distancing. The vaccine is administered in two shots. You go in for your first one, and then you have to return three weeks later for your second dose. Many people are skeptical of how Pfizer will distribute the vaccine because it is something unprecedented. Unlike a flu shot, doctors have no experience in administering this specific vaccine. We cannot also forget that the FDA must also approve the vaccine.
The scientific community assumes that they will, but we never know when a surprise may come up, after all, it is 2020.
 With the US reaching a peak of Covid-19 cases at about 150,000 cases a day, Pfizer, one of America’s largest pharmaceutical companies, held a press conference regarding their seemingly effective vaccine. They say that they have had a 90% success rate. In order for this vaccine to be available to the public, the FDA requires 2 months of safety data, which Pfizer will approach next week. They began phase 3 of their vaccine in July, and they have enrolled approximately 43,538 patients.
With the US reaching a peak of Covid-19 cases at about 150,000 cases a day, Pfizer, one of America’s largest pharmaceutical companies, held a press conference regarding their seemingly effective vaccine. They say that they have had a 90% success rate. In order for this vaccine to be available to the public, the FDA requires 2 months of safety data, which Pfizer will approach next week. They began phase 3 of their vaccine in July, and they have enrolled approximately 43,538 patients.